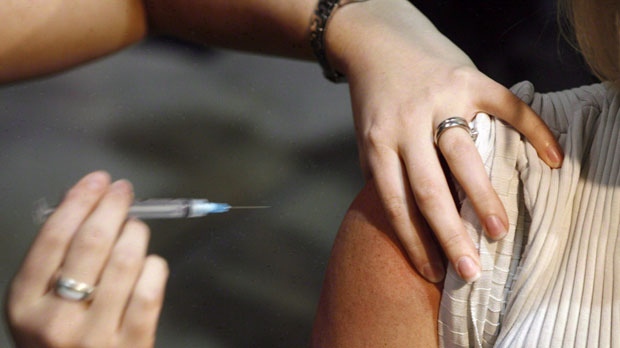
TORONTO -- Canada is following the lead of several European countries and suspending distribution of flu vaccine made by the pharmaceutical firm Novartis.
The decision relates to the discovery by the company of tiny clumps of virus particles in some batches of flu vaccines made at the Novartis production facility in Italy.
Health Canada, which announced the move, said Novartis has agreed to suspend distribution of its vaccines -- sold in Canada as Fluad and Agriflu -- while the department investigates the situation. All the Novartis vaccine Canada purchases is made at the Italian plant.
The department is also telling doctors and others who administer flu shots to hold off using Novartis product for the time being.
"We think it's prudent, given the response of certain European countries to . . . request of Novartis -- and they will be complying -- to stop distributing and then to recommend to practitioners to refrain from using the (Novartis) vaccine just until this review is completed," Dr. Paul Gully, senior medical advisory for Health Canada, said Friday.
Italy, Germany and Switzerland have suspended distribution of some Novartis flu vaccine, and in the case of Germany recalled some lots of vaccine, after the clumping issue came to light.
In a statement issued Friday night, the company said more than one million doses of its flu vaccines have been administered in Europe so far this season and no unexpected adverse events have been reported.
As well, it said that it has already delivered about 70 per cent of its Canadian order (roughly 1.5 million doses), again without hearing of problems in people who have received Novartis flu shots. The company said people who have received Novartis flu shots are not at risk.
Novartis said finding minute clumps of virus protein in vaccines is not unusual. They said their vaccines passed quality inspections and they are confident the products are safe.
"The aggregate proteins are predominantly influenza virus-derived (mainly hemagglutinin), all normal and necessary components of influenza vaccines," the company said. "Aggregation of these proteins is not unusual in vaccines manufacturing."
Hemagglutinin is the protein on the outside of flu viruses that locks onto cells in the human respiratory tract to start the process of infection. Flu vaccines are designed to provoke the immune system to produce antibodies to hemagglutinin to protect against infection.
In fact, this isn't the first time protein clumping has disrupted Canada's flu vaccine supply.
During the 2009 pandemic, there was a delay in delivery of unadjuvanted vaccine for pregnant women when GlaxoSmithKline, Canada's pandemic vaccine supplier, found visible protein aggregation in some of the vaccine.
Adjuvants are compounds that boost the response a vaccine generates. Canada used adjuvanted vaccine during the pandemic, but bought some unboosted product for pregnant women as a precaution.
Novartis makes only about 20 per cent of Canada's annual flu vaccine purchase. GlaxoSmithKline makes the bulk of Canada's seasonal flu vaccine, though a variety of other suppliers have a share of the Canadian market.
Still, because of the way vaccine orders are placed, the hold on Novartis vaccine could put some provinces and territories in a position where they face a temporary vaccine shortfall, just at the time when flu shot programs are getting underway, Gully admitted.
He said Health Canada hopes there is a rapid resolution of the situation. But if provinces or territories have a problem with supply, efforts will be made to share across jurisdictions, he said.
Late Friday night, the Saskatchewan government announced it was temporarily suspending all its influenza immunization clinics because of the Novartis situation.
"We are taking a precautionary measure while this review is underway," said Dr. Saqib Shahab, Saskatchewan's chief medical health officer.
"Our program will resume once the Health Canada review is completed."
Both Fluad and Agriflu are sold in single-dose formulations, pre-loaded into a syringe.
Fluad contains an adjuvant and is licensed for use in people 65 and older. Older adults do not mount a good response to flu vaccine and the inclusion of an adjuvant is an effort to improve the protection they get from flu shots.